Description:
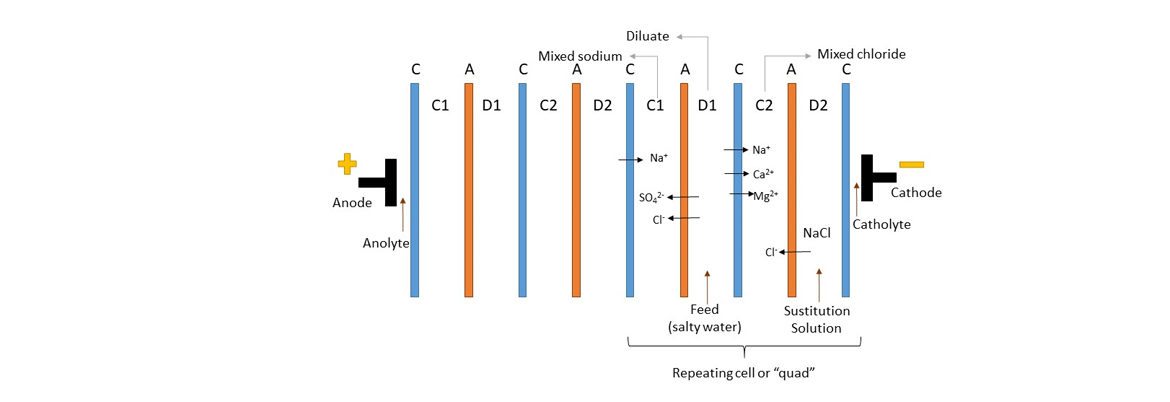
Electrodialysis Metathesis (EDM) is a variation of reversal electrodialysis in which a metathesis reaction occurs. A conventional electrodialysis (ED) system is composed of repeating cells of two alternating cation- and anion-exchange membranes. Every cell contains two compartments, one containing the feed solution and the other containing the concentrate solution. The membranes are placed between two electrodes and an electrical current passes through them to cause the mobility of the charged ions towards the corresponding electrode. In the EDM system, four alternating ion-exchange mem-branes form a repeating quad of four compartments and a substitution solution is added to provide the exchangeable ions for the metathesis reaction.
In the reaction, the feed solution, represented by MX, exchange cations and anions with the substitution solution, M′X′, to form new product salts, M′X and MX′, as represented in the equation.
MX + M′X′ -> M′X + MX′
For this purpose, two of the compartments contain the feed and a substitution solution, and are called dilute 1 (D1) and dilute 2 (D2) compartments, respectively. The other two compartments contain each the newly formed non-precipitating product salts, and are called concentrate 1 (C1) and concentrate 2 (C2) compartments.
Depending on the intended product solution in the two concentrate compartments, an appropriate substitution salt solution is fed to the D2 compartment to initiate the double decomposition reaction with the feed solution from the D1 compartment. Salts formed in the C1 compartment are predominantly rich in anions and poor in cations from the D1 compartment. Salts formed in the C2 compartment are predominantly rich in cations and poor in anions from the D1 compartment. When sodium chloride is added as the substitution solution, the C1 compartment contains mixed sodium salts, and the C2 compartment contains mixed chloride salts.
The ion exchange membranes, which are conductive and selective, allow the double exchange of ions between the saline solutions to produce concentrated salt water in addition to clean water.
Objective:
Metathesis electrodialysis is used to further concentrate brines from seawater or brackish water. With it, a greater amount of fresh water is achieved without causing the problem of scaling.
Type of water:
Brine from brackish water desalination.
Brine from seawater water desalination.
Saline wastewater.
Applications:
Zero liquid discharge.
Brine treatment and concentration.
Recovery of second generation products.
Comparison to established technologies:
Advantages:
Absence of applied pressure.
Lower fouling potential due to lack applied pressure.
Multiple application.
High water recovery: up to 80%.
Minimal maintenance compared reverse osmosis.
Provides a significant advantage in treating RO concentrate because the membrane-fouling potentials of typical scalants such as CaSO4 and CaCO3 do not increase with recovery, as is the case with RO, nanofiltration, and other forms of electrodialysis, such as electrodialysis reversal (EDR).
Two separate brine streams rich in different compounds such as Ca+2/Mg+2 or SO42- are obtained, which can be used later as by-products.
Disadvantages:
Necessary to add NaCl current to achieve equilibrium in the mass balance.
Low technology maturity status.
Applicability:
TRL 4 in 2021
Technological readiness level (TRL), it is an indicator of maturity of a particular technology. TRL is measured on a scale from 1 to 9, 1 being the lowest and 9 being the highest. A TRL of 9 reflects the implementation of an actual system in operational environment.
J.C. Mankins, Technology Readiness Levels. White Paper, April, 1995 https://www.spacepropulsion.org/uploads/2/5/3/9/25392309/john_mankins_paper_of_4-6-95_trl.pdf
Other issues:
- Hybrid systems:
Reverse Osmosis (RO) – Electrodialysis Metathesis (EDM)
Electrodialysis Metathesis (EDM)– solar evaporation
Electrodialysis Metathesis (EDM)- compounds precipitations using chemical reagents
Energy consumption:
Specific energy consumption, as reported in literature varies, depending on feed water concentrations (2-15 kWh/m3)
Website of ZELDA project. Zero liquid discharge desalination: brine treatment based on EDM and valuable compound recovery. http://zelda.ctm.com.es/es/el-proyecto/pert-diagram_es.jpg/view
Research directions
Combine with other technologies to valorize possible waste compounds as a new raw material. Circular economy.
Data sources
- Camacho, L. M., Fox, J. A., & Ajedegba, J. O. (2017). Optimization of electrodialysis metathesis (EDM) desalination using factorial design methodology. Desalination, 403, 136-143.
- Bond, R., Batchelor, B., Davis, T., & Klayman, B. (2011). Zero liquid discharge desalination of brackish water with an innovative form of electrodialysis: electrodialysis metathesis. Water Resour. J, 63(7).
- Liu, J., Yuan, J., Ji, Z., Wang, B., Hao, Y., & Guo, X. (2016). Concentrating brine from seawater desalination process by nanofiltration–electrodialysis integrated membrane technology. Desalination, 390, 53-61.
- Zhang, Y. F., Liu, L., Du, J., Fu, R., Van der Bruggen, B., & Zhang, Y. (2017). Fracsis: Ion fractionation and metathesis by a NF-ED integrated system to improve water recovery. Journal of Membrane Science, 523, 385-393.
- Medina Collana, J. T. (2018). Optimización de parámetros de operación de un módulo de electrodiálisis metátesis para tratar concentrados de agua salobre procedente de Osmosis Inversa.
- Chen, Q. B., Ren, H., Tian, Z., Sun, L., & Wang, J. (2019). Conversion and pre-concentration of SWRO reject brine into high solubility liquid salts (HSLS) by using electrodialysis metathesis. Separation and Purification Technology, 213, 587-598.
- Ahmed, F. E., Hashaikeh, R., & Hilal, N. (2020). Hybrid technologies: The future of energy efficient desalination–A review. Desalination, 495, 114659.